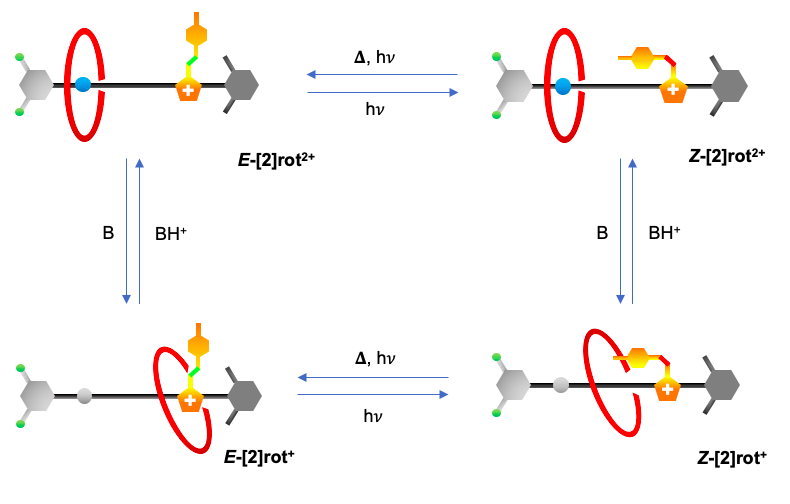
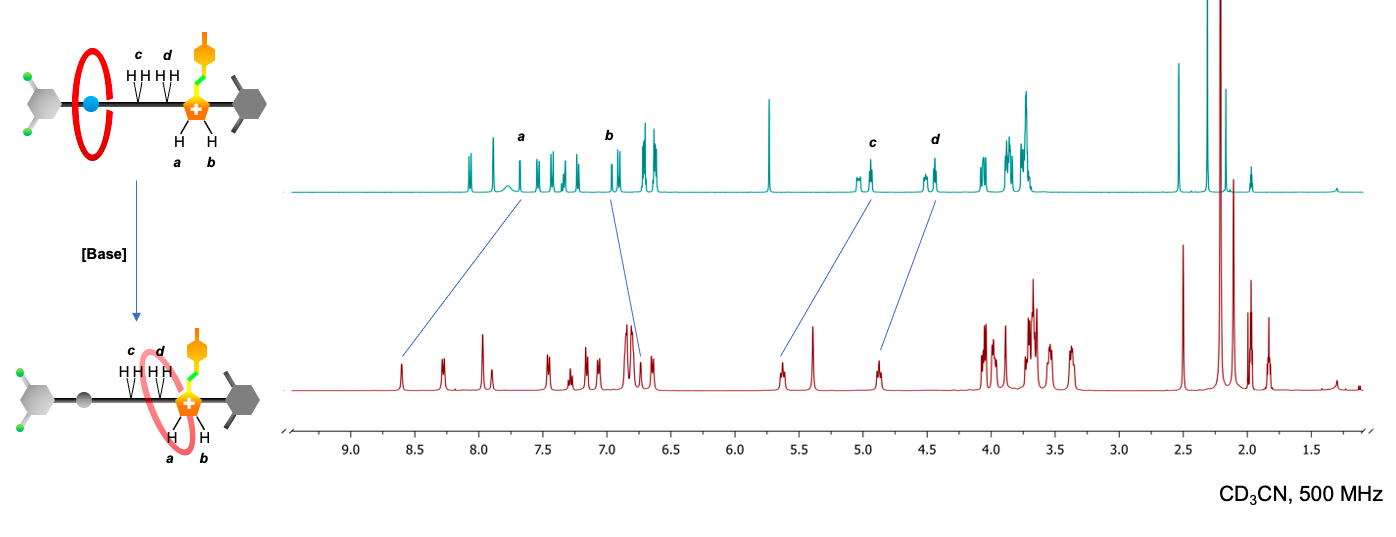
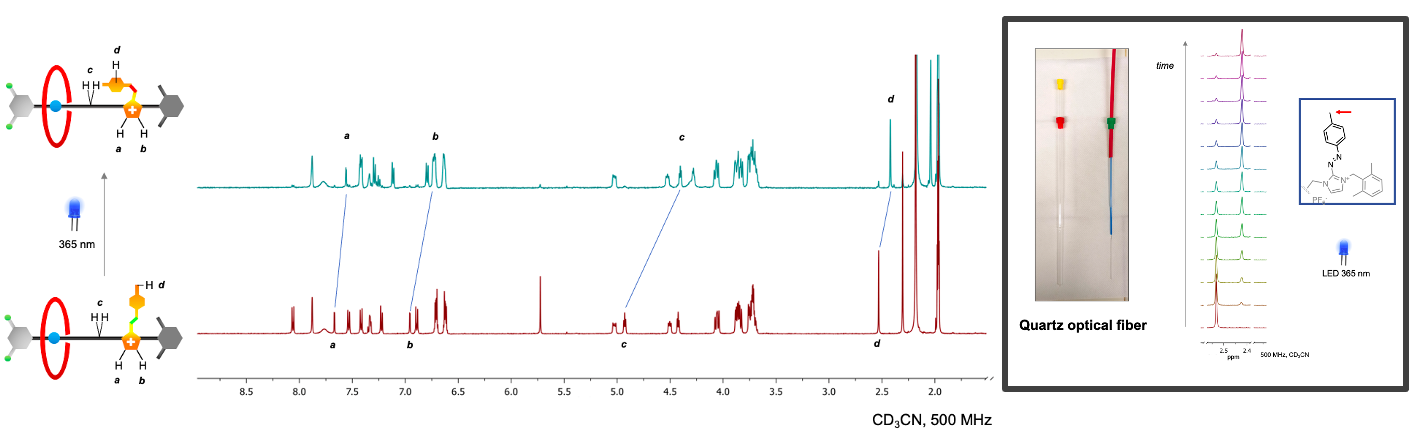
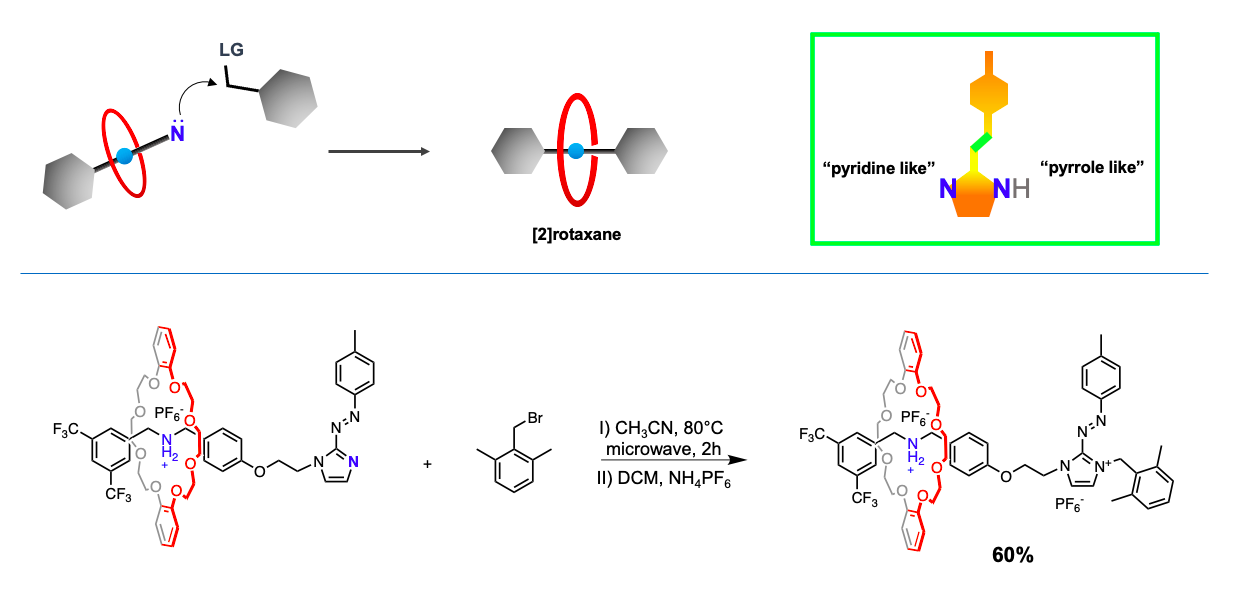
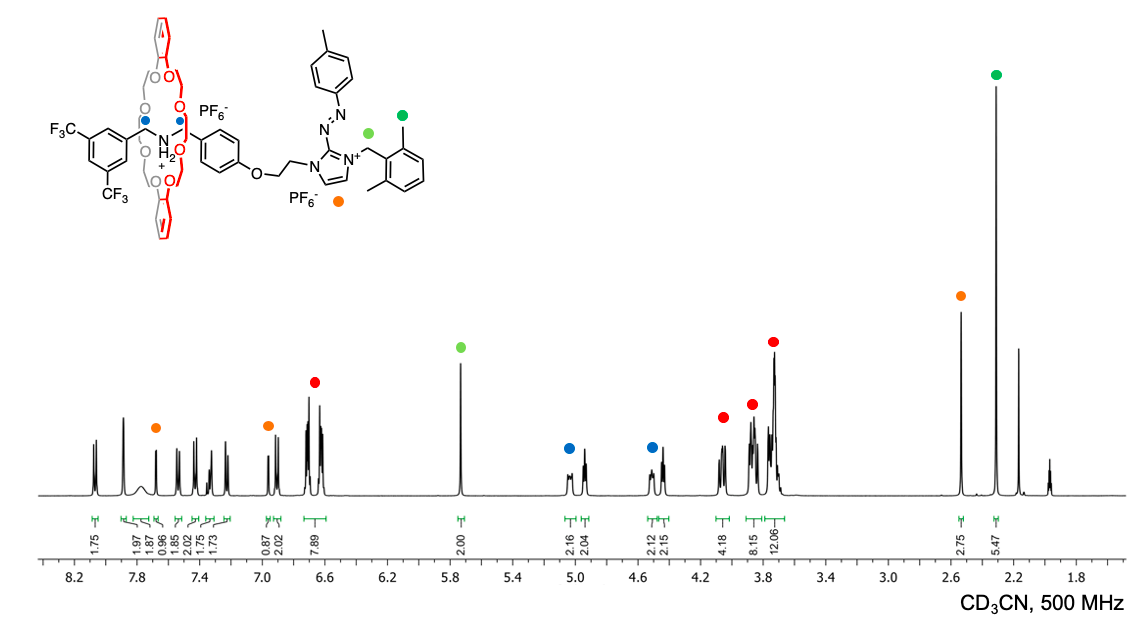
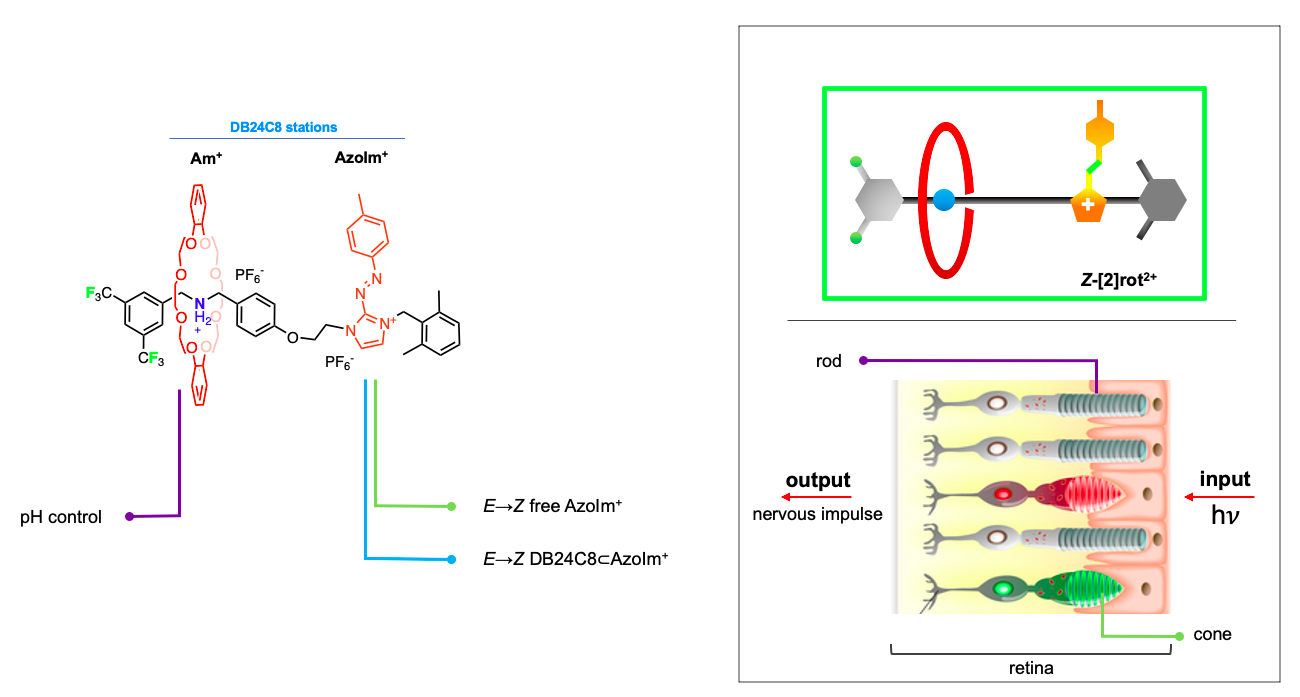
The reported rotaxane ([2]rot2+) is composed of a stronger ammonium station (Am+) and a novel, weaker phenyl azoimidazolium station (AzoIm+).
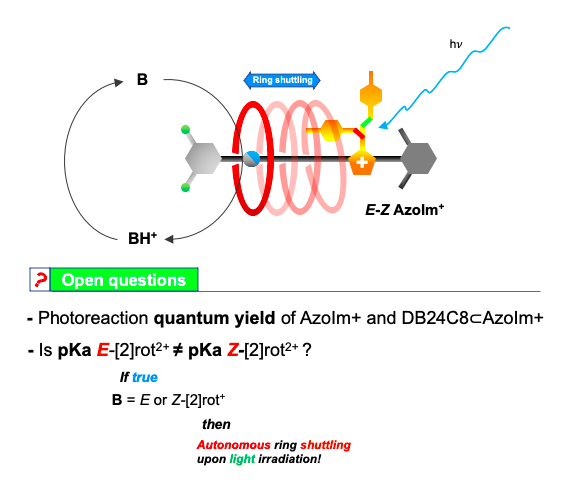
As a recent work1 shows, chemical information can be transferred upon different sites of a designed [2]rotaxane axle. This phenomenon known in several natural regulatory mechanisms as allostery, was made possible in this case thanks to the interlocked nature of rotaxanes.
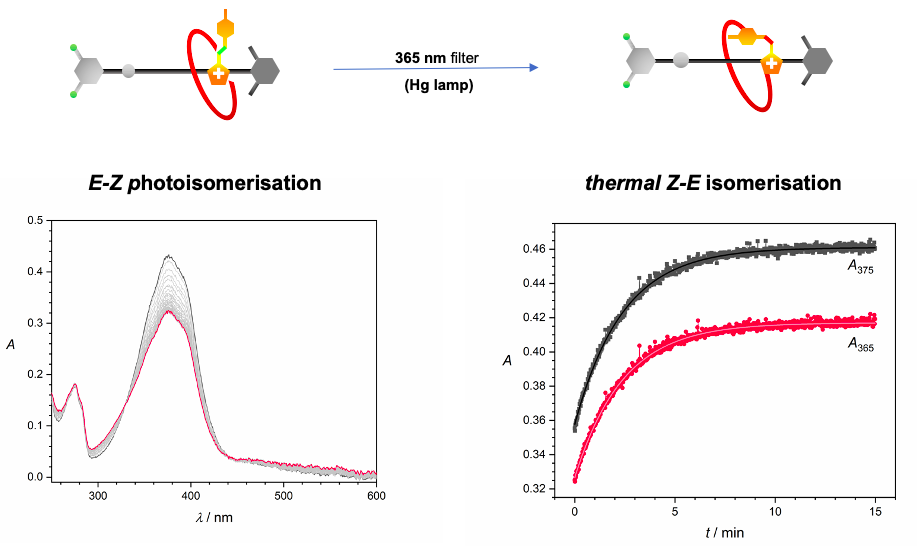
The isomerisation of the photochromic unit caused by light (INPUT) in the [2]rot2+ could be responsible for the modification of the thermodynamic properties of the Am+ station (OUTPUT). The process presents an analogy with the animal vision mechanism in which light captured by cone and rod cells is transduced into a nervous impulse directed to the brain.